Alarming: Resistances against a new active-substance combination detectable prior to use in Germany
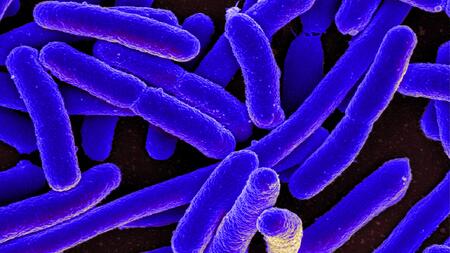
The rod-shaped Escherichia coli bacterium can cause severe infections (colorized scanning electron micrograph).
Enterobacteria such as Escherichia coli or Klebsiella pneumoniae are dreaded hospital germs that can cause serious intestinal and urinary tract infections. The risk of infection with these pathogens is growing, because they are becoming increasingly resistant worldwide and thus resistive to a group of antibiotics that effectively constitutes the critical reserve for emergencies: carbapenems. A new combination of two antimicrobial substances was seen as a beacon of hope for the treatment – but this hope has now been dashed by a recent study at Justus Liebig University Giessen (JLU) and the German Center for Infection Research (DZIF). Even before the substances were ever used in Germany, the scientists were able to find bacteria that this new combination could no longer touch.
In 2017, the World Health Organization (WHO) issued a list of twelve multidrug-resistant pathogens for which new active substances are urgently needed. The highest priority is given to Acinetobacter baumannii and Pseudomonas aeruginosa as well as the enterobacteria mentioned earlier that are resistant to carbapenems. For them, a new combination of two antimicrobial substances – aztreonam and avibactam – is seen as a beacon of hope.
Beacon of hope against specific resistances
“Aztreonam avibactam is a combination of an older antibiotic – aztreonam – with a newer inhibitor – avibactam – that can void the effectiveness of the resistance to carbapenems, thus rendering the bacteria vulnerable again,” explains Dr Can Imirzalioglu, DZIF scientist at the JLU Institute of Medical Microbiology and co-author of the study. Good efficacy is expected especially against enterobacteria, whose mechanism of resistance is based on a particular type of carbapenemase enzymes. So far, infections with such carbapenem-resistant pathogens are often very difficult to treat, as the treatment options with existing antibiotics are severely limited or even no longer exist,” the researcher adds.
At present, the fixed combination of these two substances is not yet freely available as a therapy and is only used in clinical studies. Within Europe, Greece and Italy are among countries setting their hopes on the combination of aztreonam and avibactam, as this type of carbapenemase is more widespread there than in Germany. However, the incidence of carbapenem-resistant pathogens is steadily increasing across the world. Against this backdrop the team led by resistance researcher Prof. Dr Patrice Nordmann at the University of Fribourg (Switzerland) has been able to identify mechanisms resulting in resistance to aztreonam-avibactam. Furthermore, they have already detected pathogens with these properties. Dr Yancheng Yao, co-author of the study and DZIF scientist at the Institute of Medical Microbiology at JLU Giessen, describes these mechanisms: “This resistance arises from a specific combination of changes in existing structures of the bacteria in connection with the acquisition of certain resistance genes.”
Study detects early resistances in Germany
In cooperation with the scientists from Fribourg, the Giessen-based research group has now been able to detect such pathogens in Germany for the first time by means of a genome analysis of bacteria conducted as part of a surveillance study for highly resistant pathogens in Hesse (SurvCARE). “This is worrying, because this active-substance combination is not yet used clinically here,” explains Prof. Dr Trinad Chakraborty, Director of the Institute of Medical Microbiology at JLU and co-coordinator of the research area Healthcare-Associated and Antibiotic-Resistant Bacterial Infections at the DZIF. “It is also alarming that this resistance has also been detected in bacteria carrying carbapenemases that are very common in Germany. As a result, the potential of aztreonam-avibactam as an initial therapy is significantly limited. Its expectation as a combination of hope in infections with carbapenem-resistant pathogens may not be fulfilled,” the scientist concludes.
The study, which has now been published in the journal Antimicrobial Agents and Chemotherapy, also points to the importance of a genome-based surveillance of such pathogens. “The genome-based analysis of highly resistant pathogens is not only important for the analysis of outbreak situations or transmission events, when used continuously, it also fulfils an important guard function for the early detection of new resistance developments and emerging pathogens,” emphasizes Prof. Dr Linda Falgenhauer, co-author of the study, DZIF scientist and professor of Genome-based Surveillance and Epidemiology of Multidrug-resistant Pathogens at the JLU Institute of Hygiene and Environmental Medicine. The collected data were easy to be exchanged in digital form in collaborations in order to make important developments of supra-regional or international relevance speedily visible.
The study once again underlines the importance of developing new antibiotics, a focal point at the DZIF. The DZIF is a founding member of the recently formed INCATE initiative, which aims to fill the pipeline for new drugs.