The development of a vaccine against MERS virus gets international support
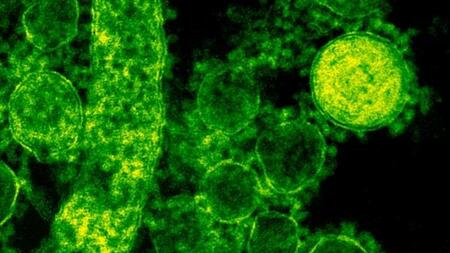
MERS Corona virus particles outside the cell
CEPI (the Coalition for Epidemic Preparedness Innovations) supports the development and manufacture of a vaccine against Middle East Respiratory Syndrome Coronavirus (MERS-CoV), a zoonotic disease that is spread to humans by dromedary camels. The promising vaccine candidate was advanced by the German Center of Infection Research (DZIF) up to clinical phase I. Next development activities will now be performed by IDT Biologika and a MERS-CoV Vaccine Consortium of scientific and clinical organizations, including DZIF.
First identified in 2012, MERS-CoV is carried by camels and spread to humans by droplet infection of the airways. Infection can cause respiratory disease including fever and cough that can progress into severe pneumonia. About 35% of reported patients with MERS-CoV have died and the WHO has highlighted MERS-CoV as a priority disease for research and development in view of its epidemic potential. No vaccine is currently available.
The vaccine candidate from DZIF is based on a recombinant Modified Vaccinia Ankara (MVA) vector with an inserted MERS gene. IDT Biologika developed a proprietary cell substrate for large-scale production of the MVA based vaccine. The first generation of the MVA-MERS vaccine is being assessed in an ongoing clinical trial at DZIF. The publication of first results is prepared.
About MERS-CoV Vaccine Consortium
The development activities will be performed by a MERS-CoV Vaccine Consortium of IDT Biologika, DZIF, Viroscience Department at the Erasmus Medical Center, and the clinical research organization CR2O.
About CEPI
CEPI is an innovative partnership between public, private, philanthropic and civil organizations founded in Davos in 2017 to develop vaccines to stop future epidemics. Since its launch in January 2017, CEPI has announced two Calls for Proposals. The first was for candidate vaccines against MERS-CoV, Nipah and Lassa viruses. The second call demanded the development of platforms that can be used for rapid vaccine development against unknown pathogens. Learn more at cepi.net.
About IDT Biologika
IDT Biologika is an innovative, privately-held company with nearly 100 years of experience in researching, developing, manufacturing and marketing products for the global protection of human and animal health. The company produces vaccines and pharmaceuticals according to the highest quality standards, for its own animal health products and under contract. Further information can be found on the website of IDT Biologika at www.idt-biologika.com.
More information