Research focus on antimicrobial resistence
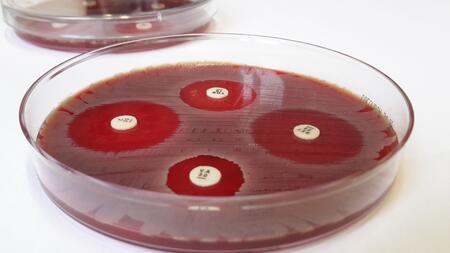
The agar diffusion test makes antibiotic resistance visible.
The rapid spread of antibiotic-resistant bacteria is calling infection researchers to action. Which mechanisms protect bacteria and which methods can make them vulnerable once again? In an article published 2011 in "Science", researchers postulated that sulphide protects bacteria against different antibiotics. However, using a new technique, DZIF scientist Fabian Grein has now shown that such a “universal” resistance mechanism does not exist.
Fabian Grein and his research group at the University of Bonn researched the effect of sulfide on the sensitivity of Staphylococcus aureus to the most important classes of antibiotics. He heads the DZIF junior research group which focuses on anti-staphylococcal targets and consequently tested the “sulfide postulation” on staphylococci, a group of dreaded hospital pathogens. What is the underlying mechanism for sulfide’s assumed protective function? And could it be targeted specifically to consequently break down bacterial resistance?
Does sulfide universally protect bacteria against antibiotics?
In order to investigate the effect of sulfide on bacteria, the scientists initially had to develop a modified agar diffusion test that permits continuous incubation with sulfide. Sulfide oxidates rapidly and this is the only method to counteract this. Using this system, the staphylococci were tested systematically and compared to the most important antibiotics.
“With our technique we were able to show that sulfide solely protects bacteria from aminoglycosides,” explains Fabian Grein. Aminoglycosides are a heterogeneous group of antibiotics that disturb bacterial protein production, which is fatal for bacteria. Important members are gentamicin, streptomycin and kanamycin.
To date, it has been assumed that sulfide reduces oxidative stress, which is thought to occur during all antibiotic treatments. DZIF scientists from the research field “Healthcare-associated and Antibiotic-resistant bacterial Infections” can now refute this postulation. “Aminoglycosides are the only antibiotics that require energy to pass through the bacterial membrane in order to reach their target,” explains Fabian Grein. Together with his group, he has now been able to show that addition of sulfide partially poisons cells. Subsequently, insufficient energy is available for uptake of aminoglycoside antibiotics, which consequently protects the cells. This mechanism is similar to that used by so-called small colony variants (SCV), a subpopulation of S. aureus that tend to develop during chronic persistent infections. As a result of mutations, SCVs have lower membrane potentials, which reduces their uptake of aminoglycosides and makes them resistant to this group of antibiotics.
Surprising results
Fabian Grein admits that he himself was surprised to have refuted the longstanding postulation that sulfide provides universal protection. “Additionally, we have seen that the amount of sulfide produced varies strongly between different types of bacteria and is too low to provide effective protection in Staphylococcus aureus.”
Grein and his team now intend to continue searching for new structures that could be used to target staphylococci. “Of course, it would have been nice to identify sulfide as the culprit and use it as a basis for developing a universal strategy against antibiotic resistance.” Nonetheless, the team has already started working on new target structures. The focus here is on target sites that enable killing a specific pathogen or to manipulate the sites in such a way that sensitivity to classic antibiotics is re-established.
The paper was recently recommended by the post publications peer review service F1000Prime and was ranked as “exceptional”.