Nucleoside Booster Project: Broad-spectrum antivirals for pandemic preparedness
The SARS-CoV-2 pandemic and other epidemics in recent decades have highlighted the need to develop effective measures to combat emerging infectious diseases in a timely manner. For most emerging viral pathogens, no vaccines or antiviral drugs are yet available. Since it is difficult to predict which virus or viral variants might cause an outbreak in the future, drugs that exert broad antiviral activity are particularly needed.
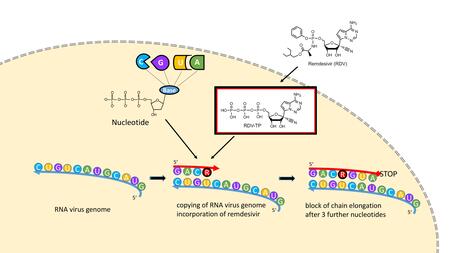
In the Nucleoside Booster Project, DZIF scientists from Heidelberg University Hospital and other DZIF member institutions are working together with the Drugs for Neglected Diseases initiative (DNDi) on the identification of chemical compounds with a broad antiviral spectrum of activity in order to prepare for future viral outbreaks. This requirement is met by the nucleoside analogues, which are among the particularly successful chemical classes in antiviral therapy. These substances, which resemble natural DNA and RNA building blocks, include the nucleosidic inhibitors used in antiviral therapy against HIV or HBV infection and the nucleoside analogue remdesivir (see figure below). Nucleoside analogues are incorporated into the genome copy during viral genome replication, but are chemically modified in such a way that either genome replication stops (like for remdesivir) or the newly formed genomes contain so many mutations that they are no longer functional (like for the SARS-CoV-2 drug molnupiravir). Because of this property and the fact that viruses have great difficulty in developing resistance to these drugs, they are promising starting substances for the development of broad-spectrum antivirals.
A collection of nucleoside analogue compounds was selected and acquired by DNDi, an organisation aiming to develop easily accessible drugs for impoverished populations, to be subjected to antiviral testing in the laboratory. The Antiviral Compound Testing Platform (ACTP), established by DZIF and located at several DZIF member and associated institutions, is available for testing. ACTP provides various in vitro test systems based on cell lines and primary cells, as well as in vivo test systems in rodents. In addition, the platform comprises a unique collection of highly pathogenic viruses, including coronaviruses (SARS-CoV, MERS-CoV), filoviruses (Ebola), bunyaviruses (RVFV, CCHF), flaviviruses (ZIKV, DENV, WNV), influenza viruses, arenaviruses (Lassa) and hantaviruses. Among these are nine of the ten viruses classified by WHO as causative agents of priority diseases, which have the potential to cause public health emergencies and for which there are no effective countermeasures.
The goal of the scientists in the Nucleoside Booster Project is now to use the ACTP test platform to identify those nucleoside analogues in the DNDi collection of nucleoside analogue compounds that exhibit the broadest possible antiviral activity and can serve as potential leads for the development of therapeutics.