Preclinical development of Amidochelocardins
The challenge of antibiotic drug development today is the discovery and optimisation of novel bioactive compounds that do not show cross-resistances with clinically applied antibiotics, ideally due to a novel mode of action (MoA) which adresses unexploited microbial targets. In the case of chelocardin, this antibiotic substance was identified several decades ago and is referred to as “atypical tetracycline” due to structural similarities to the known class of antibiotics, which act as inhibitors of bacterial protein synthesis. However, chelocardin shows unique features regarding its chemistry and bioactivity and displays at least a dual MoA, which is directed against unknown target(s) in the bacterial membrane. Recent studies could improve both the potency and stability of the compound by genetic engineering of its biosynthetic pathway, resulting in the novel derivative amidochelocardin, which is now undergoing structural optimisation to generate a lead candidate for preclinical studies. Due to its pharmaceutical properties the compound is developed to ultimately treat urinary tract infections, especially those caused by multidrug-resistant uropathogens.
Background
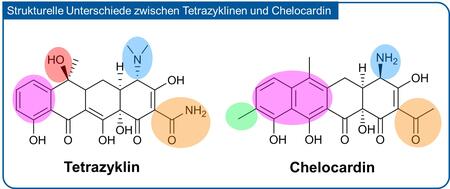
Chelocardin was isolated and described in the early 1960s as a molecule with broad-spectrum antibiotic activity, and despite its similarities to the class of tetracyclines it was found that it acts according to a yet unknown antimicrobial mechanism. Importantly, the compound has a strong potency in killing various Gram-negative pathogens, which belong to the major global health threats, especially if they carry multiple resistances against currently used antibiotics. Some of these dangerous microbes are listed by the World Health Organization (WHO) as high priority pathogens. The natural product chelocardin is produced by the nonpathogenic soil bacterium Amycolatopsis sulphurea and can be isolated at relatively high levels under optimized laboratory cultivation conditions (more than 1 gram of substance per litre of bacterial culture). Its biosynthesis as well as the corresponding biosynthetic gene cluster, encoding a highly complex enzyme machinery called “polyketide synthase”, were described only recently.
In spite of the sparse knowledge about its detailed Mode of Action, Abbot Laboratories (USA) used the substance already in the 1970s to conduct a small clinical phase II study with hospitalised patients suffering from kidney infections in Zagreb, Croatia. In this study, chelocardin showed very promising results in a cohort of 12 patients. However, the drug was apparently not considered as a candidate for further therapeutic development during this era when antibiotic resistance was not regarded as major health threat.
Structural improvement of the compound
With the increasing need for novel antimicrobial therapeutics and due to the promising activity of chelocardin as well as the lack of cross-resistances with known antibiotics, scientists from the Helmholtz Centre for Infection Research (HZI) decided to recover the neglected molecule in order to create novel chelocardin analogues with improved bioactive properties. Part of this program is done in collaboration with the industrial partner AciesBio and the research group of Prof. H. Petkovic at University of Ljubljana, Slovenia, who provided expertise in producer strain genetics and cultivation as well as compound isolation.
From chelocardin to amidochelocardin
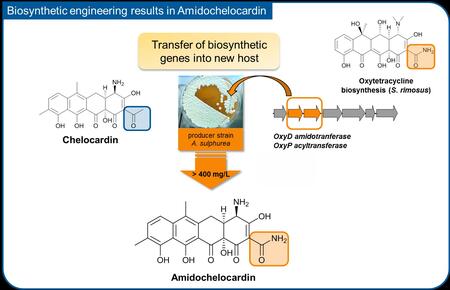
An important step towards the structural improvement of the compound was made recently: The biosynthesis of the chelocardin molecule was genetically engineered by introducing genes from a classical tetracyline biosynthetic pathway into the Amycolatopsis strain. The result of this bioengineering process was a kind of hybrid molecule combining structural features from both the chelocardin and the oxytetracycline scaffold, and was called “amidochelocardin”. Strikingly, the novel compound displayed enhanced bioactivity against several priority pathogens, and was more stable in solution over a longer period of time, which is an important pharmaceutical property required for the efficacy of treatment in patients.
The molecule on its way to the clinic
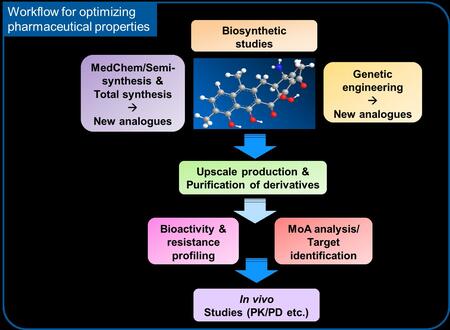
Since 2016 the ongoing pharmaceutical improvement of amidochelocardin is supported by the DZIF, which allowed the funding of a collaborative translational development project within the research field “Novel Antibiotics” in order to bring the molecule on its way to the clinic. At present, four different HZI research groups located in Braunschweig and Saarbrücken are busy optimising different features of the amidochelocardin molecule in relation to its bioactivity profile, a process which is called SAR profiling. They also aim to improve the production and purification of the compound and of further structural analogues which are generated by chemical modification of the (amido)chelocardin scaffold, and also to decipher the MoA by various physiological and biochemical approaches. Animal studies already showed efficacy in mouse infection models, and additional series of pharmacological data are acquired e. g. by using mouse models at HZI or with external partners in order to optimise the efficacy parameters of the lead structures for upcoming preclinical studies. The indication for envisaged therapeutic treatment remains basically unchanged to the indication that was already found in the early clinical study in 1977, which is the utilisation of the novel drug for curing complicated urinary tract infections, in particular when associated with multidrug-resistant bacteria such as ESBL (extended-spectrum-beta-lactamases)-producing enterobacteria, which cannot be treated with most current antibiotics.